
What is ILB®?
ILB is a modified form of dextran sulfate, a type of sugar molecule. It was discovered serendipitously when performing studies on another indication and turned out to have a pleiotropic mechanism of action, stimulating release of multiple growth factors with a wide range of downstream effects.
Neurodegenerative conditions
Neurodegenerative conditions are disorders that progressively damage the nervous system, particularly the brain. These diseases often lead to cognitive decline, motor dysfunction, and other neurological impairments. Some of the most common neurodegenerative conditions include:
- Alzheimer’s disease – Characterized by memory loss, confusion, and cognitive decline due to the buildup of amyloid plaques and tau tangles in the brain.
- Parkinson’s disease – A movement disorder caused by the degeneration of dopamine-producing neurons, leading to tremors, stiffness, and balance issues.
- Huntington’s disease – A genetic disorder that affects movement, cognition, and psychiatric health, resulting from a mutation in the HTT gene.
- Amyotrophic lateral sclerosis (ALS) – Also known as Lou Gehrig’s disease, ALS leads to the progressive loss of motor neurons, causing muscle weakness and paralysis.
- Multiple sclerosis (MS) – An autoimmune condition where the immune system attacks the protective myelin sheath around nerves, disrupting communication between the brain and body.
While there is no cure for most neurodegenerative diseases, treatments focus on managing symptoms and slowing progression. Research continues to explore new therapies, including genetic interventions and neuroprotective strategies.
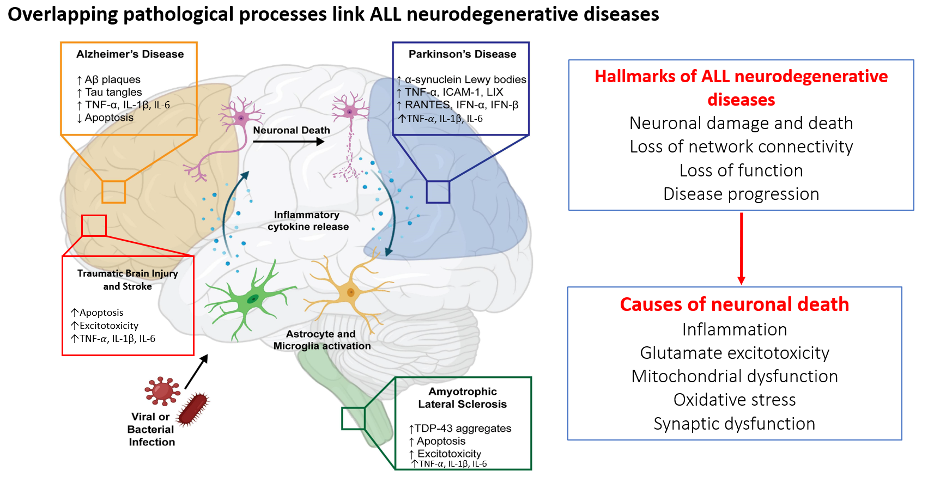
Image courtesy of Prof Ann Logan, Univ of Warwick, UK
Preclinical studies
ILB® has been investigated in several preclinical studies in glaucoma, stroke, acute limb ischemia and traumatic brain injury (TBI). In general, ILB seems to have wide regenerative and neuroprotective effects in all these conditions. ILB has also been demonstrated to prevent viral infections of cells in an in vitro environment.
The mechanism of action of ILB has been extensively studied in cells, animals and humans. In summary, ILB seems to have pleiotropic effects, triggering release of growth factors, e.g. HGF and BDNF, that are bound to glypican in blood-vessel walls. ILB also downregulates the inflammatory response and prevents fibrosis, possibly by upregulating decorin expression. This action initiates a cascade of transcriptional, metabolic, and immunological changes that mitigate key pathological features of neurodegeneration—including oxidative stress, mitochondrial dysfunction, glutamate excitotoxicity, and inflammation.
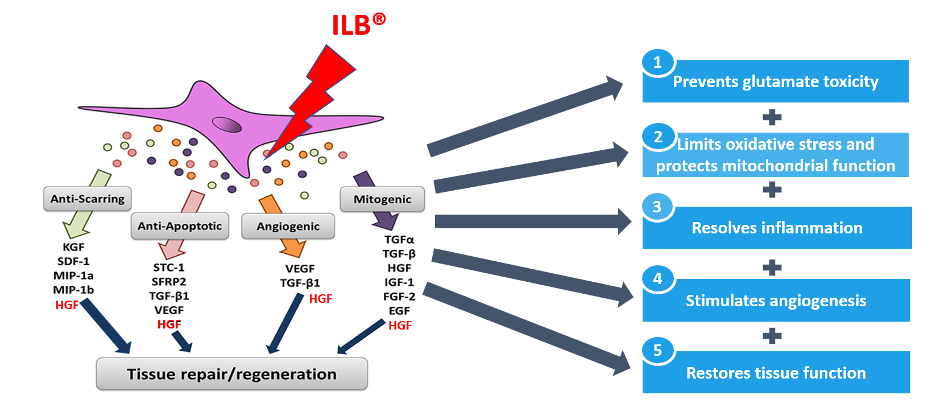
Image courtesy of Prof Ann Logan, Univ of Warwick, UK
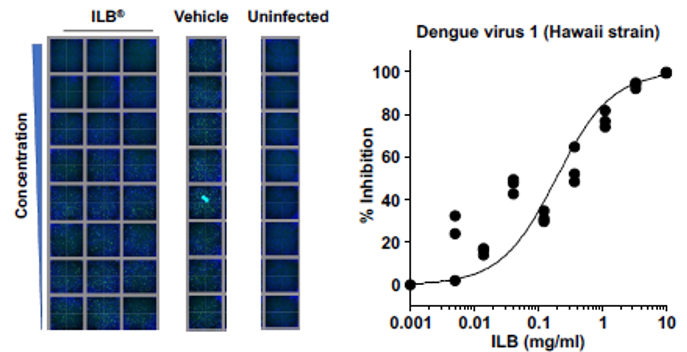
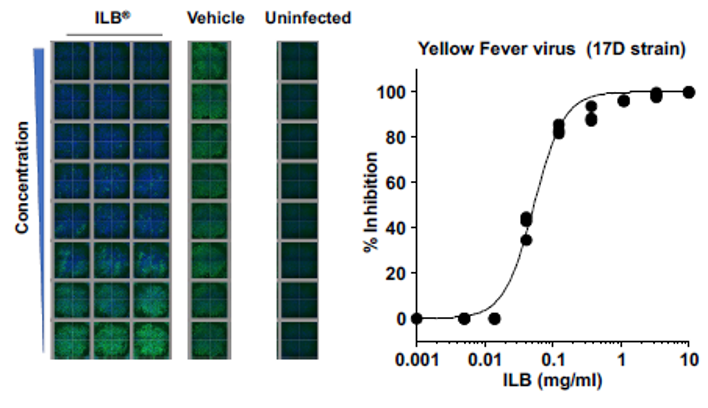
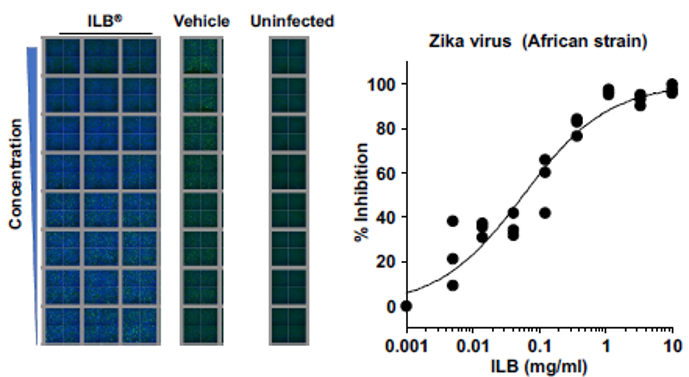
ILB inhibits viral infection of human cells
ILB has been demonstrated in laboratory experiments to inhibit the infection of human cultured cells by Dengue, Zika, and Yellow Fever viruses. Sample results:
References
Logan A et al., 2022. The mechanism of action of a novel neuroprotective low molecular weight dextran sulphate: New platform therapy for neurodegenerative diseases like Amyotrophic Lateral Sclerosis. Front. Pharmacol. 13:983853. doi: 10.3389/fphar.2022.983853
Logan A et al, 2022. A clinical stage LMW-DS drug inhibits infection of human cells by Dengue, Zika and Yellow Fever viruses. bioRxiv preprint doi: 10.1101/2022.08.31.503293
>> ILB in glaucoma
>> ILB in TBI
>> ILB in stroke
>> ILB in limb ischemia
References
Mechanism of action:
Logan A, et al, 2022. Front. Pharmacol. 13:983853. doi: 10.3389/fphar.2022.983853
Glaucoma:
Hill LJ et al, 2021. npj Regenerative Medicine 6:3. doi: 10.1038/s41536-020-00110-2
TBI:
Lazzarino G et al, 2020. Antioxidants 9:850. doi: 10.3390/antiox9090850
Lazzarino G et al, 2022. Int. J. Mol. Sci. 23:8460. doi: 10.3390/ijms23158460
Viral infection:
Logan A et al, 2022. bioRxiv preprint doi: 10.1101/2022.08.31.503293
ALS:
Lazzarino G et al, 2021. J. Pers. Med. 11:794. doi: 10.3390/jpm11080794
Logan A et al, 2022. PLoS ONE 17: e0267183. doi: 10.1371/journal.pone.0267183.
Srinivasan V et al, 2024. PLoS ONE 19: e0291285. doi.: 10.1371/journal.pone.0291285
